建議您使用以下瀏覽器觀看本網站,
以獲得最佳瀏覽效果。
Corporate News
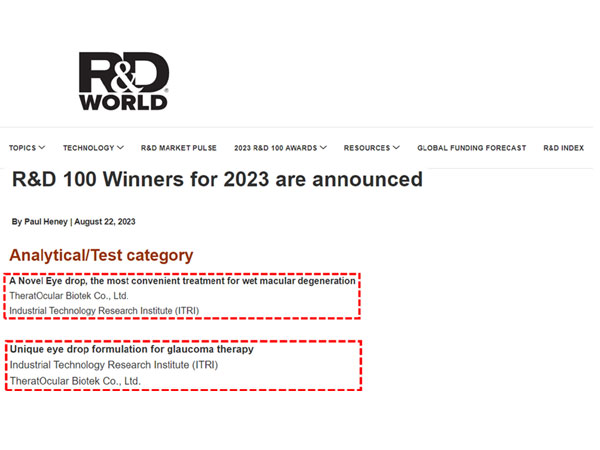
TO-O-1001 & TO-O-1002 won the 2023 R&D 100 Award
2023.08.22

MG-O-1002 (TO-O-1002) project won the 2023 Edison AwardsTM
2023.04

ITRI Special Issue ----
Interview with Dr. Sean Chen, chairman of TheratOcular Biotek
2022.12

New blue ocean for ophthalmology drugs; Taiwanese manufacturers are actively improving innovative technologies
2022.11.24

Old formula New amazing effect, TheratOcular Biotek makes it easy to treat eye diseases - Executive VP Simon Huang Interview
2022.11.24

63th Annual Meeting of Ophthalmology & 15th Asia Pacific Vitreoretinal Society (APVRS) Congress
2022.11.18

Press conference of innovative eye drops
2022.11.08

TO-O-1002 Investigator meeting (IM) in Thailand
2022.09.22

The phase II clinical trial of TO-O-1002 approved by the Central Research Ethics Committee (CREC) in Thailand on 11 Aug 2022.
2022.08.15

The phase I/II clinical trials of TO-O-1001 approved by the Australian Human Research Ethics Committee (HREC) on 4 Aug 2022.
2022.08.09

The company Chair, Dr. Sean Chen, and the SAB Chair, Dr. Lu Da-Wen, get together with all the honorary members of the Company Scientific Advisory Board (SAB) in a banquet.
2022.08

TheratOcular Team meet up with its RD/BD partner, the ITRI-BEL team, at the 2022 Asia Biotech Exhibition.
2022.07


